ETHICS REVIEW COMMITEE
Latest Updates
About the CNU Ethics Review Committee
About the CNU Ethics Review Committee
The CNU Ethics Review Committee (CNU ERC) was approved by the Board of Regents on December 7, 2017 through Resolution No. 117 series of 2017. Subsequently, the approval of the CNU ERC Standard Operating Procedures, Special Guidelines and Ethical considerations (Resolution No. 14 series of 2018) was given on March 8, 2018. The CNU ERC is designated to approve, monitor, and review biomedical and behavioral research involving humans and all forms of life with the aim of protecting the rights and welfare of the research subjects.
The Board shall have the following functions:
a. Require all faculty members and students who will conduct research that involves human participants to submit a complete description of the proposed research using the CNU ERC Protocol Submission Overview;
b. Review all researches submitted by faculty members and students that involve human participants to assure, both in advance and by periodic review, that appropriate steps are taken to protect the rights and welfare of humans
participating as subjects in a research study:
c. Assess the ethics of the research and its methods, to promote fully informed and
voluntary participation by prospective subjects who are themselves capable of making such choices (or, if that is not possible, informed permission given by a suitable proxy) and to maximize the safety of subjects once they are enrolled in the project; and
d. Approve or disapprove the implementation of the research project or withhold approval of the implementation of the study pending modifications or changes to protocol or the consent procedures.
Ethical Considerations
The CNU Ethics Review Committee (ERC) is dedicated to assisting student and faculty researchers in ensuring the ethical integrity of their research protocols. By adhering to its ethical guidelines, the ERC ensures that research conducted under its purview is ethically sound, respects the rights and dignity of participants, and maintains the highest standards of academic integrity. To uphold high ethical standards, the ERC requires all submitted research protocols to include provisions addressing the following ethical considerations:
- Voluntary Participation:
Participants should not be coerced through bribery, pressure, or blackmail into participating in research. Every participant must have the autonomy to choose to be involved in the study and must retain the right to withdraw at any time without any repercussions. - Anonymity and Confidentiality:
Anonymity involves ensuring that participants remain unidentifiable and untraceable, while confidentiality pertains to the non-disclosure of collected data to unauthorized individuals or entities. Research protocols must clearly outline the methods for collecting, storing, processing, and disposing of data to safeguard participants’ identities and privacy. - Conflict of Interest:
A conflict of interest arises when personal interests could compromise an individual’s judgment or decision-making during the research process. Researchers must explicitly declare any potential conflicts of interest to maintain transparency and integrity in their work. - Informed Consent or Assent:
Participants of legal age (18 years and older) must provide informed consent, whereas minor participants (under 18 years) must provide assent, accompanied by a child information sheet and a consent form from their parents or legal guardians. The informed consent form should include a brief description of the study, its objectives, the extent of participant involvement, potential risks and benefits, and contact information for the primary investigator. - Risks:
Researchers must outline the potential harms that participants may face if they choose to participate in the study. These risks can be physical, social, psychological, or legal. Identifying potential risks allows researchers to minimize their impact and to develop solutions and support mechanisms should these risks materialize. - Benefits:
Participants may incur personal expenses or face inconveniences during data collection. Therefore, they should be justly compensated when applicable. For instance, traveling to and from an interview location may require reimbursement for transportation and food expenses. Additionally, if the data collection impedes on the productive economic hours of the participants, the researchers must give compensation to the participants equivalent to the number of hours they have allotted for the study.
Instructions for Primary Investigators Regarding Protocol Submission
Before you submit your protocols for research ethics review, read the instructions below carefully and follow each one strictly. Failure to follow these instructions will result in the rejection of your protocol and it will not be included for presentation during the ERC bimonthly meeting. Take note that the office of the ERC is processing the submissions on a first come first serve basis. Thus, returned and unprocessed submissions will go back to the end of the queue if there are lacking documentary requirements or if the instructions are not followed.
- All protocol submissions will be done online through the ERC email cnuerc@cnu.edu.ph.
- Use the latest version of the ERC Forms. The latest versions of the forms are the ones found in this page available for download.
- Only the primary investigator shall submit the protocol and other documents using his or her CNU email address.
- Use only one single email thread for each protocol. Refrain from creating another email thread for the same protocol. All subsequent submissions, replies and inquiries for the protocol will be sent in the same email thread.
- Attach your documents as PDF or word files. Do not send us your files in a google drive or link. This is to ensure that the ERC office will have continuous and unhindered access to the files at all times.
- Label your files appropriately, preferably as reflected in the list of documents below. Do not attach the same file repeatedly in varying file formats.
- Submit the approved protocol, signed compliance checklist, cover letter, and accomplished Forms 6 or 7, 8, and 9 in PDF format.
- Submit the accomplished Forms 1, 3, 4 and 5 as Word File.
- Double check if all the required information needed in each Form is provided.
- Submission of Form 9 is optional. If your protocol does not involve any human participants (studies such as but not limited to: Literature review, Systematic Review, Data Mining, etc.) or does not involve vulnerable populations (minors, PDLs, PWDs, etc.) or sensitive and intrusive topics, you may submit to us the accomplished Form 9. However, take note that this does not mean that the protocol will immediately be given an Exemption from Review decision, but rather your application for exemption will be considered during the deliberation.
- Before sending your protocol for research ethics review, check the completeness of your documents and if everything is in order.
Updated List of documentary requirements for Ethics Review Application
- Approved Proposal/ Protocol (with CV, Gantt chart, research instruments) (PDF)
- Signed Compliance Checklist (PDF) for Thesis and Dissertation; or
Certificate of Evaluation and approval signed by the Advisor for non-thesis and non-dissertation; or
Technical Review Board Approval from RDP for Faculty Research
All of the following documents must be submitted to ERC for protocol ethics review. Make sure that your submission is complete. All forms (except for the approved manuscript and compliance checklist) are provided by the ERC office.
1. ERC FORM 1 – Documentary Receipt Form (Word)
2. ERC FORM 2 – Cover Letter (PDF)
3. ERC FORM 3 – Protocol Review Application Form (Word)
4. ERC FORM 4 – Protocol Review Assessment Form (Word)
5. ERC FORM 5 – Informed Consent Form Checklist (Word)
6. ERC FORM 6 – Informed Consent Form (for Participants of legal age) (PDF)
7. ERC FORM 7 – Child Information Sheet and Assent Form (for participants who are minors) (PDF)
8. ERC FORM 8 – Detailed Budget Sheet (PDF)
9. ERC FORM 9 – Application for Exemption Form (PDF) (Optional)
Updated List of documentary requirements for Ethics Clearance Application
- Copy of the Approved Completed Manuscript (PDF)
- Signed Compliance Checklist or Approval Sheet (PDF)
- ERC FORM 10 – Progress report (PDF)
- ERC FORM 11 – Reportable Negative Event From (RNEF) (PDF)
- ERC FORM 12 – Study Completion / Final Report (PDF)
Updated List of documentary requirements for Protocol Amendments
- Request letter justifying the protocol amendments (signed by the adviser/ panel members) (PDF)
- Revised protocol or informed consent form (PDF)
- ERC FORM 13 – Protocol Amendment Form (PDF)
Other Forms
- ERC FORM 14 – Premature Termination/ Suspension/ Discontinuation Report Form (PDF)
Ethics Review Committee Members (2nd Semester SY2025-2026)
List of documentary requirements for Ethics Review Application
All of the following documents must be submitted to ERC for protocol ethics review. Make sure that your submission is complete. All forms (except for the approved manuscript and compliance checklist) are provided by the ERC office.
1. ERC FORM 1 – Documentary Receipt Form (Word)
2. ERC FORM 2 – Cover Letter (PDF)
3. ERC FORM 3 – Protocol Review Application Form (Word)
4. ERC FORM 4 – Protocol Review Assessment Form (Word)
5. ERC FORM 5 – Informed Consent Form Checklist (Word)
6. ERC FORM 6 – Informed Consent Form (for Participants of legal age) (PDF)
7. ERC FORM 7 – Child Information Sheet and Assent Form (for participants who are minors) (PDF)
8. ERC FORM 8 – Detailed Budget Sheet (PDF)
9. ERC FORM 9 – Application for Exemption Form (PDF) (Optional)
Downloadable Forms
- 1. ERC FORM 1 - Documentary Receipt Form (Word)
- 2. ERC FORM 2 - Cover Letter (PDF)
- 3. ERC FORM 3 - Protocol Review Application Form (Word)
- 4. ERC FORM 4 - Protocol Review Assessment Form (Word)
- 5. ERC FORM 5 - Informed Consent Form Checklist (Word)
- 6. ERC FORM 6 - Informed Consent Form (for Participants of legal age) (PDF)
- 7. ERC FORM 7 - Child Information Sheet and Assent Form (for participants who are minors) (PDF)
- 8. ERC FORM 8 - Detailed Budget Sheet (PDF)
- 9. ERC FORM 9 - Application for Exemption Form (PDF) (Optional)
- ERC FORM 10 - Progress report (PDF)
- ERC FORM 11 - Reportable Negative Event From (RNEF) (PDF)
- ERC FORM 12 - Study Completion / Final Report (PDF)
- ERC FORM 13 - Protocol Amendment Form (PDF)
- ERC FORM 14 - Premature Termination/ Suspension/ Discontinuation Report Form (PDF)
- FORM 1 Protocol review application form
- FORM 2 Protocol review assessment form
- FORM 3 Informed consent form checklist
- FORM 4 Participant information sheet and informed consent
- FORM 5 Child information sheet and assent form
- FORM 6 Detailed budget sheet
- FORM 7 Document receipt form
- FORM 8 Exemption from Review Application
- FORM 9 Reportable Negative Event Form (RNEF)
- FORM 10 Premature, Suspension, Discontinuation Report Form
- FORM 11 Study Completion/ Final Report Form
- FORM 12 Assessment of resubmitted protocol
- FORM 13 Protocol Amendment Form
- FORM 14 Premature Termination/ Suspension/ Discontinuation Report Form
- FORM 1 Protocol review application form
- FORM 2 Protocol review assessment form
- FORM 3 Informed consent form checklist
- FORM 4 Participant information sheet and informed consent
- FORM 5 Child information sheet and assent form
- FORM 6 Detailed budget sheet
- FORM 7 Document receipt form
- FORM 8 Exemption from Review Application
- FORM 9 Reportable Negative Event Form (RNEF)
- FORM 10 Premature, Suspension, Discontinuation Report Form
- FORM 11 Study Completion/ Final Report Form
- FORM 12 Assessment of resubmitted protocol
Summary of the Ethics Review Process
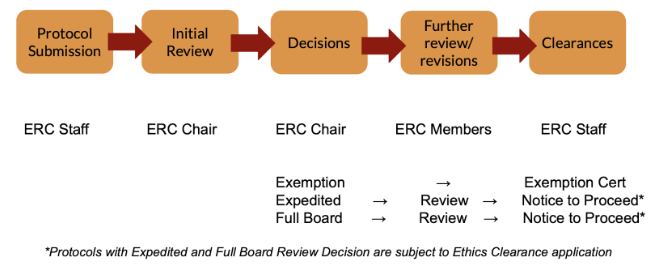
Reminders:
- The Ethics Review Committee conducts a bimonthly meeting, usually on the second and the last Fridays of the month (or 15th and 30th day of the month, whichever is applicable).
- The ERC office is following a first come first serve basis for all protocol submissions.
- Release of Exemption from review decision takes up to 20 days.
- Release of Notice to Proceed takes up to 60 days, depending on the protocol.
- The Exemption from Review Certificate will only be given to those protocols with exemption from review decision. These protocols will not anymore require the release of the Notice to Proceed and Ethics Clearance.
- Notice to Proceed (NTP) is only given to those protocols with expedited and full board decisions. The NTP gives the researchers the go signal to start their data gathering.
- The NTP is different from Ethics Clearance. The clearance will only be given after the completion and approval by the technical panel committee and the submission of documentary requirements.
- The processing of payment for ethics review will be done after a decision has been made for your protocol submission. The ERC will endorse you to the accounting office and the payment instructions will be sent to you by them through email. You can pay physically through the university teller or through online payment.
Processing fee:
Undergraduate – No payment
Masters – P1,500.00
Doctoral – P2,000.00
Faculty research without RDP review – P2,000.00
Contact Information
Rhoniel Ryan J. Ymbong, M.Sc.
Chair, Ethics Review Committee
For more inquiries and online submissions, contact us via:
Email: cnuerc@cnu.edu.ph
Office: 3rd Floor, TAC Building, Cebu Normal University-Main Campus, Osmeña Blvd., Cebu city
Visit our website at www.cnu.edu.ph to access the downloadable forms for ethics review application and the citizen’s charter.













